

Barred Tiger Salamander. Photograph by Carl S. Lieb.
Larvae are often sold as commercial live bait in the Southwestern U.S. as "waterdogs," although this term is properly associated with other types of salamanders elsewhere in the United States. The species occupying the Chihuahuan Desert Region of Texas and eastern to central New Mexico currently is known as the Barred Tiger Salamander, Ambystoma mavortium, but has been considered as a subspecies of A. tigrinum until recently. Populations in western New Mexico are usually referred to as A. tigrinum nebulosum, the Arizona Tiger Salamander. The taxonomic situation is complicated because of a) polymorphisms over much of the species range (Collins et al. 1980), b) introductions due to release of bait animals in many parts of the non-native range in the Southwest, and c) the presence of a genetically distinct taxon, the Sonoran (Huachuca) Tiger Salamander (A. t. stebbinsi, see Jones et al., 1988) in the San Rafael Valley of southeastern Arizona.
The most recent reviews of the species (Schaffer and McKnight, 1996; Irschick and Schaffer, 1997) suggest that the Tiger Salamanders inhabiting the Sierra Madre Occidental, as well as arid areas south of the Chihuahuan Desert region proper in Mexico, represent at least one or more distinct species (combined into "Ambystoma velasci" at present).
Transformed adults may be as small as 15.5 cm in total length (Webb and Roueche, 1971), but are often larger and up to 24 cm. The dorsal coloration of adults often consists of yellow bars or elongated pale spots on the back on a dark brown to black ground color. Nevertheless, uniform olive or reticulated transformed adults are occasionally encountered. Larval coloration is typically a uniform drab olive brown, olive green, or yellowish brown, rarely whitish-pale (Webb and Roueche, 1971; Fernandez and Collins, 1988). Ground coloration of both adults and larvae may be influenced by environmental substrate and age (Fernandez and Collins, 1988). Larvae and neotenic adults (see Natural History) reach at least 26 cm in total length. These aquatic forms have conspicuous external gills and enlarged tail fins that are lacking in the transformed adults. Recently transformed adults may show stubby gill elements in the process of being resorbed.
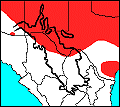
The Tiger Salamander is widespread in temperate central North America, including southern Canada throughout the Great Plains and eastern Rocky Mountains to the northern Chihuahuan Desert. It is also present but more spottily distributed in the eastern United States from the Ohio Valley to the Atlantic Seaboard and Southeastern States. The distribution of tiger salamanders in northern Mexico is poorly understood.
As a species, this salamander occupies a wide variety of habitats ranging in elevation from sea level to over 10,000 feet and from boreal forest to desert scrubland. Nevertheless, populations throughout the geographic range are often spottily distributed and are probably sensitive to the reliable presence of suitable aquatic habitats at the appropriate times of year.
Like several members of its salamander family (Ambystomatidae), Tiger Salamanders exhibit facultative metamorphosis, whereby environmental influences stimulate larvae to develop into sexually mature adults with aquatic rather than terrestrial adaptations (Duellman and Trueb, 1986; Pough et al., 2001). This retention of larval characteristics (e.g., external gills, tail with fin margin) is accomplished through a developmental process called paedomorphosis; the form paedomorphosis apparently takes in salamanders involves neoteny, whereby the heterochronous development of the somatic tissues of immature animals proceeds apace slower than the development of the gonads and other components of the reproductive system (Pough et al., 1999). Facultative metamorphosis is highly adaptive in arid regions where terrestrial conditions outside of the immediate vicinity of the breeding ponds are inimical to terrestrial salamanders. Chihuahuan Desert populations of Tiger Salamanders are no exception, and thus terrestrial transformed adults may be seldom seen in a region that supports a large but relatively unobvious population of neotenic adults and larvae in local cattle ponds and other impoundments. Heavy rainstorms occasionally bring transformed individuals out onto roads at night.
Transformed terrestrial adults typically feed on insects and other arthropods. The larvae likewise feed on arthropods that live or fall into the water, spadefoot toad (Scaphiopus) larvae, and each other. A neotenic "cannibal morph" (Rose and Armentrout, 1976) may also appear in populations; this form consisting of individuals with larger heads, wider mouths, and more effective predatory teeth. Cannibal morphs eat a variety of vertebrate and invertebrate animal prey besides other salamanders (Hammerson, 1982; Collins, 1982); the environmental cues that result in their appearance in Tiger Salamander populations are uncertain, but have been tied to larval density (Collins and Cheek, 1983) and aquatic pathogens (Pfennig et al. 1991).
The principal biological studies of the Tiger Salamander in the Chihuahuan Desert are of a population at Taylor Well on the Jornada Experimental Range of New Mexico by Webb (1969), Webb and Roueche (1971), and Whitford and Massey (1970). The Taylor Well was revisited by Michael Ross of UTEP in 1988 and the population found to be still extant.
Fertilization is internal by spermatophores; the courtship behavior takes place in water (see Arnold, 1976, for full description; summary in Duellman and Trueb, 1986). Eggs are laid in temporary ponds and hatch into aquatic larvae. Webb and Roueche's (1971) Jornada study indicated that egg deposition occurs when summer rains fill the pond, but may also occur in the spring and fall if sufficient rainfall occurs during these periods, and probably occurs on occasion throughout mild winters. In the Webb and Roueche's Jornada site, eggs are typically attached singly or in pairs to objects in the water (e.g., submerged twigs); the larvae at hatching were 9 to 10 millimeters in total length.
Larval Ambystoma tigrinum are regionally used as live bait in some southwestern states and, except in Arizona, for the most part the harvest of larvae and adults from the wild for commercial or other purposes is essentially unregulated. The exception is in the San Rafael Valley of Arizona, where the Sonora Tiger Salamander (Ambystoma t. stebbinsi, also called the Huachuca Tiger Salamander) is listed as an Endangered Species by the U.S. Fish and Wildlife Service. This group of populations breed in livestock ponds; the putative natural breeding habitats no longer exist. Principal threats to the Sonoran Tiger Salamander include a) introduced non-native predators in the form of bullfrogs, crayfish, and fish, b) genetic swamping with other forms of tiger salamanders released as unwanted live bait by anglers into the populations, and c) infectious disease in the form of a highly lethal iridiovirus (Jancovich, et al. 1997). There is also accumulating evidence that other populations of Tiger Salamanders may also be susceptible to such viruses, and these can effect mass mortalities of larvae leading to local population declines and/or disappearances (Daszak, P. et al. 1999). A detailed recovery plan for the Sonora Tiger Salamander has been drafted and is now in the process of being reviewed for approval (U.S. Fish and Wildlife Service, 1999).
Arnold, S. J. 1976. Sexual behavior, sexual interference, and sexual defense in the salamanders Ambystoma maculatum, Ambystoma tigrinum, and Plethodon jordani. Zeit. Tierpsychol. 42:247-300.
Collins, 1982. Amphibians and reptiles in Kansas. Second Edition. Univ. Kansas Mus. Nat. Hist. Publ. Educ. Seri. (8):1-356.
Collins, J. P., J. B. Mitton, and B. A. Pierce. 1980. Ambystoma tigrinum: a multispecies conglomerate? Copeia 1980(4):938- 941.
______, and J. E. Cheek, 1983. Effects of food and density on development of typical and cannibalistic salamander larvae in Ambystoma tigrinum nebulosum. American Zool. 23(1):77- 84.
Conant and Collins, 1991. [on general Chih. herp ref list]
Daszak, P., L. Berger, A. A. Cunningham, A. D. Hyatt, D. E. Green, and R. Speare. 1999. Emerging Infectious Diseases 5(6):1-21 [online journal: http://www.cdc.gov/ncidod/EID/vol5no6/daszak.htm
Degenhardt, W. G. et al. 1996 [on general Chih. herp ref list]
Dixon, J. R. 2000. [on general Chih. herp ref list]
Duellman, W. E. and L. Trueb. 1986. Biology of amphibians. McGraw-Hill Book Company, New York.
Fernandez, P. J. and J. P. Collins, 1988. Effect of environment and ontogeny on color pattern variation in Arizona tiger salamanders (Ambystoma tigrinum nebulosum Hallowell). Copeia 1988(4):928-938
Gehlbach, F. R. 1967. Ambystoma tigrinum. Catalog. American Amphibs. Rept. 52.1-.4.
Hammerson, G. A. 1982. Amphibians and reptiles in Colorado. Colorado Div. Wildl. Publ. DOW-M-I-27-82:1-131.
Homomuzki, J. R. , and J. P. Collins. 1987. Trophic dynamics of a top predator Ambystoma tigrinum nebulosum in a lentic community. Copeia 1987(4):949-957.
Irschick, D. J. and H. B. Shaffer. 1997. The polytypic species revisited: Morphological differentiation among tiger salamanders (Ambystoma tigrinum) (Amphibia: Caudata). Herpetologica 53(1):30-49.
Jancovich, J. K., E. W. Davidson, J. F. Morado, B. L. Jacobs, and J. P. Collins. 1997. Isolation of a lethal virus from the endangered tiger salamander, Ambystoma tigrinum stebbinsi. Diseases of Aquatic Organisms 31:161-67.
Jones, T. R., J. P. Collins, T. D. Kocher, and J. B. Mitton. 1988. Systematic status and distribution of Ambystoma tigrinum stebbinsi Lowe (Amphibia: Caudata): Copeia 1988: 621-635.
Pierce, B., and J. B. Mitton. 1980. Patterns of allozyme variation in Ambystoma tigrinum mavortium and A. t. nebulosum. Copeia 1980(3):594-605.
Pfenning, D. W., M. L. G. Loeb, and J. P. Collins. 1991. Pathogens as a factor limiting the spread of cannibalism in tiger salamanders. Oecologia 88(2):161-166.
Pough, F. H., C. M. Janis, and J. B. Heiser. 1999. Vertebrate Life, Fifth Edition. Prentice Hall, Upper Saddle River, New Jersey.
Pough, F. H., R. M. Andrews, J. E. Cadle, M. L. Crump, A. H. Savitzky, and K. D. Wells. 2001. Herpetology, 2nd edition. Prentice-Hall, Upper Saddle River, New Jersey.
Rose, F. L. and D. Armentrout. 1976. Adaptive strategies of Ambystoma tigrinum (Green) inhabiting the Llano Estacado of west Texas. J. Anim. Ecol. 45(3):713-729.
Schaffer, H. B. 1983. Biosystematics of Ambystoma rosaceum and A. tigrinum in northwestern Mexico. Copeia 1983:67-78.
Schaffer, H. B. and M. McKnight. 1996. The polytypic species revisited: Genetic differentiation and molecular phylogenetics of the tiger salamander(Ambystoma tigrinum) (Amphibia: Caudata) complex. Evolution 50:417-433.
Stebbins, 1985. [on general Chih. herp ref list]
U.S. Fish and Wildlife Service. 1999. Sonora tiger salamander (Ambystoma tigrinum stebbinsi) draft recovery plan. U.S. Fish and Wildlife Service, Phoenix, Arizona. iv + 90 pp.
Webb, R. G. 1969. Survival adaptations of tiger salamanders (Ambystoma tigrinum) in the Chihuahuan Desert. Pp. 143-147, In C. C. Hoff and M. L. Riedesel (eds.), Physiological systems in semiarid environments. Univ. New Mexico Press, Albuquerque. xi + 239 pp.
Webb, R. G., and W. L. Roueche. 1971. Life history aspects of the tiger salamander (Ambystoma tigrinum mavortium) in the Chihuahuan Desert. Great Basin Nat. 31(4):193-212.
Whitford, W. G., and M. Massey. 1970. Responses of a population of Ambystoma tigrinum to thermal and oxygen gradients. Herpetologica 26(3):372-376.
Account by: Carl S. Lieb and Randy Powell (comment on recent nomenclature by A.H. Harris)
Last Update: 5 Apr 2009